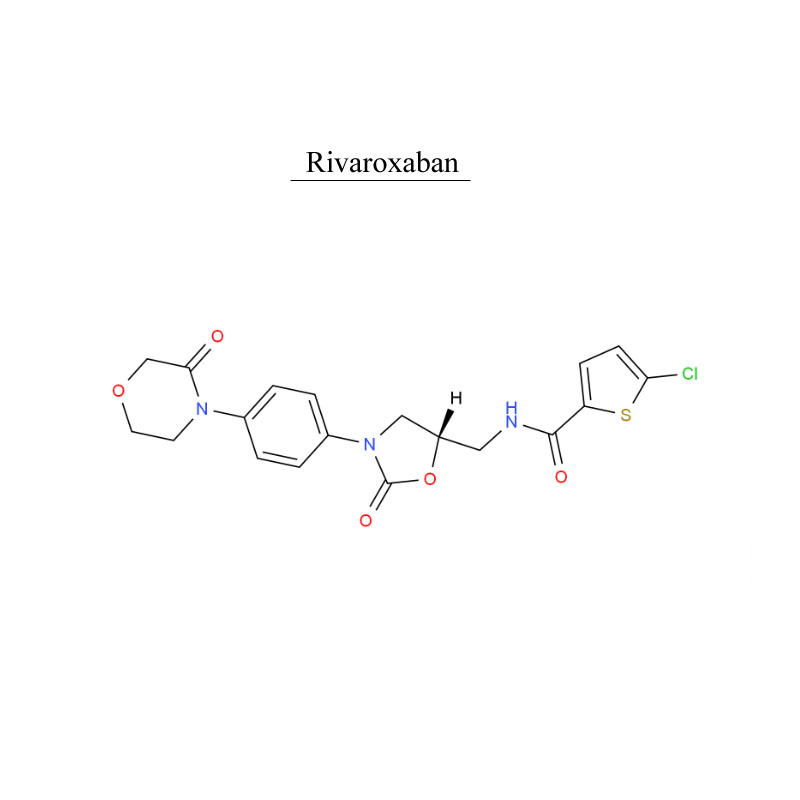
Rivaroxaban 366789-02-8 systema san- guine antithrombosis
Mercedem:T/T, L/C
Product Origin:China
Shipping Portus:Beijing/Shanghai/Hangzhou
Facultas productio;200kg / mensis
Order(MOQ);25kg
Tempus ducere:III Days
Repono conditione:Condita in loco frigido, sicco, cella temperie.
Sarcina materia:tympanum
Sarcina amplitudo:25kg/orum
Salus notitia:Non periculosum bonis
Introductio
Rivaroxaban, medicamentum anticoagulantis est (sanguis tenuior).Utitur in adultis fibrillationibus atrialibus non valvulae (praeter fibrillationem atrialem propter morbum cordis valvulare rheumaticum, et fibrillationem atrialem post cor valvae repositum) ad periculum plagae et embolismi systemici redigendum.
Rivaroxaban, sanguinem clots tractare et prohibere.
Rivaroxaban adhibetur f pro aegris adultis coxam electivam subeundae vel genu chirurgiae repositum ne venarum thrombosis.
Specificatio (EP)
| Item | Specification |
| Aspectus | Album aut flavo pulveris |
| Solubilitas | In aqua fere insolubilis, in DMSO libere solubilis, in ethanolo anhydroso et in heptanis fere insolubilis. |
| Lepidium sativum | IR: Imaginis parere debet cum WS |
| HPLC-RT specimen castitatis enantiometricae sub experimento parendum cum WS. | |
| Aquae | ≤0.5% |
| Cinis sulphate | ≤0.1% |
| Damnum in siccitate | ≤0.5% |
| Metalla gravis | ≤20ppm |
| Enantionmer | immunditia A: ≤0.4% |
| RELICTUM solvendo | Ethanol: ≤5000ppm Ethyl acetas: ≤5000ppm Acetone: ≤5000ppm N,N-Dimethylformamide: ≤880ppm Methylene chloride: ≤600ppm Benzene: ≤2ppm |
| Substantia cognata | Impuritas B: ≤0.10% Impuritas D: ≤0.10% Immunditia E: ≤0.10% Impuritas F: ≤0.10% G: ≤0.10% Impuritas H: ≤0.10% Immunditia: ≤0.10% Impuritas J: ≤0.10% RVB-4-NJSH: ≤0.10% RVB-ZA: ≤0.10% RVB-BAM: ≤0.10% RVB-ZC: ≤0.10% RVB-HBY: ≤75ppm Alia immunditia ignota: ≤0.10% Totalis immunditiae: ≤0.3% |
| Magnitudo particula distribution | D10:/ D50:/ D90:/ |
| Assay (Anhydrous substantia) | 98.0-102.0 |